Improper cable gland selection in medical and cleanroom environments causes contamination risks, regulatory compliance failures, and equipment malfunctions that result in patient safety hazards, costly facility shutdowns, and FDA violations, while inadequate sealing, inappropriate materials, and poor cleanability lead to bacterial growth, particulate contamination, and sterility breaches that compromise critical healthcare operations. Many facility managers struggle with selecting glands that meet stringent medical standards while maintaining reliable electrical performance.
Selecting cable glands for medical equipment and cleanrooms requires understanding FDA regulations, USP Class VI1 material requirements, IP68+ sealing standards, and cleanroom classification needs, with medical-grade stainless steel or specialized polymer construction providing biocompatibility, chemical resistance, and smooth surfaces for effective cleaning and sterilization while maintaining electrical integrity in critical healthcare applications. Success depends on balancing regulatory compliance with operational reliability.
Having worked with hospital engineers across major medical centers in Boston, pharmaceutical manufacturers in Switzerland, and cleanroom facilities throughout Singapore, I’ve learned that medical-grade cable glands are essential for maintaining sterile environments and ensuring patient safety. Let me share the critical knowledge for selecting optimal glands for your medical and cleanroom applications.
Table of Contents
- What Makes Medical Cable Glands Different from Standard Glands?
- How Do You Meet FDA and Medical Device Regulations?
- What Materials Are Required for Cleanroom Applications?
- How Do You Ensure Proper Cleaning and Sterilization Compatibility?
- What Are the Key Selection Criteria for Different Medical Applications?
- FAQs About Medical Cable Glands
What Makes Medical Cable Glands Different from Standard Glands?
Medical cable glands differ from standard glands by using biocompatible materials, featuring smooth cleanable surfaces, meeting FDA regulations, providing enhanced sealing for contamination prevention, and offering chemical resistance to cleaning agents and sterilization processes, with specialized designs that eliminate crevices where bacteria could harbor while maintaining electrical performance in critical healthcare environments.
Understanding these differences is crucial because medical applications have unique safety and regulatory requirements that standard industrial glands cannot adequately address.
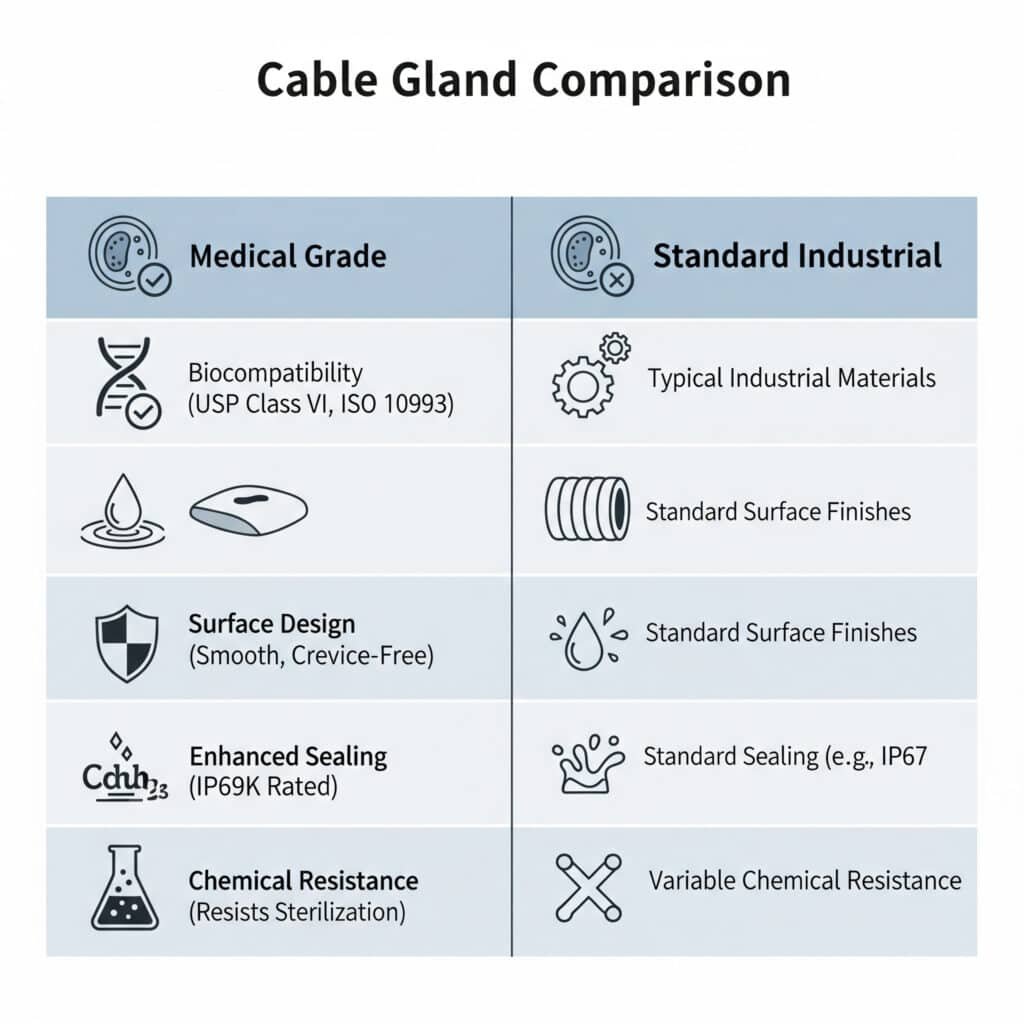
Biocompatibility Requirements
USP Class VI Compliance: Medical cable glands must use materials that pass United States Pharmacopeia Class VI biological testing for biocompatibility and safety in medical device applications.
ISO 10993 Standards: Materials must meet ISO 109932 biological evaluation standards for medical devices, ensuring no cytotoxic, sensitizing, or irritating effects on human tissue.
FDA 21 CFR 177 Approval: Food-grade materials meeting FDA regulations for direct and indirect food contact are often required for pharmaceutical and medical device applications.
Biocompatible Polymers: Specialized materials like medical-grade PEEK, PTFE, or pharmaceutical-grade nylons provide biocompatibility without compromising mechanical properties.
Surface Design for Cleanability
Smooth Surface Finish: Medical glands feature ultra-smooth surfaces with Ra values typically below 0.8 micrometers to prevent bacterial adhesion and enable effective cleaning.
Crevice-Free Design: Elimination of sharp corners, deep threads, and complex geometries that could harbor bacteria or resist cleaning and sterilization processes.
Rounded Edges: All external surfaces feature rounded edges and smooth transitions to facilitate cleaning and prevent damage to cleanroom gloves or sterile packaging.
Minimal Surface Area: Streamlined designs minimize surface area exposed to contamination while maintaining necessary mechanical and electrical performance.
Enhanced Sealing Performance
IP68+ Ratings: Superior environmental protection beyond standard IP68, often achieving IP69K for high-pressure, high-temperature cleaning applications.
Contamination Barriers: Multiple sealing stages prevent ingress of particles, bacteria, and cleaning chemicals that could compromise sterile environments.
Pressure Resistance: Enhanced pressure ratings to withstand aggressive cleaning procedures and sterilization processes without seal failure.
Long-Term Reliability: Sealing materials maintain integrity through repeated sterilization cycles and exposure to harsh cleaning chemicals.
Chemical Resistance Properties
Cleaning Agent Compatibility: Resistance to hospital-grade disinfectants, quaternary ammonium compounds, hydrogen peroxide, and other aggressive cleaning chemicals.
Sterilization Resistance: Materials withstand gamma radiation, ethylene oxide, steam autoclaving, and other sterilization methods without degradation.
Pharmaceutical Chemicals: Resistance to solvents, acids, bases, and pharmaceutical compounds commonly found in medical manufacturing environments.
Temperature Stability: Maintain properties through sterilization temperature cycles and cleanroom temperature control requirements.
David, a facilities manager at a major pharmaceutical manufacturing plant in New Jersey, faced recurring contamination issues in their sterile filling lines where standard cable glands were harboring bacteria despite rigorous cleaning protocols. The existing brass glands had complex thread patterns and surface finishes that couldn’t be adequately sterilized, leading to batch rejections and FDA compliance concerns. We specified medical-grade stainless steel cable glands with electropolished surfaces and crevice-free designs that met USP Class VI requirements. The upgrade eliminated contamination sources, achieved 99.9% cleaning effectiveness, and helped the facility pass FDA inspections while reducing product rejection rates by 85%. 😊
How Do You Meet FDA and Medical Device Regulations?
Meeting FDA and medical device regulations requires using materials with proper certifications, maintaining detailed documentation, following Good Manufacturing Practices (GMP)3, ensuring traceability, and implementing quality systems that comply with 21 CFR Part 820, with medical cable glands requiring FDA-approved materials, biocompatibility testing, and manufacturing controls that ensure consistent quality and safety in medical device applications.
Regulatory compliance is non-negotiable in medical applications because failures can result in patient harm, product recalls, and severe legal consequences.
FDA Material Requirements
21 CFR Part 177: Materials in contact with pharmaceuticals or medical devices must meet FDA food additive regulations for indirect food contact substances.
Master Access File (MAF): Suppliers must maintain FDA Master Access Files documenting material safety, manufacturing processes, and quality control procedures.
Drug Master File (DMF): For pharmaceutical applications, materials may require Drug Master File registration with detailed composition and manufacturing information.
Certificate of Compliance: Suppliers must provide certificates confirming materials meet all applicable FDA regulations and specifications.
Medical Device Quality Standards
ISO 13485 Compliance: Manufacturing must follow ISO 13485 medical device quality management systems for design, production, and post-market surveillance.
21 CFR Part 820 (QSR): Quality System Regulation compliance ensuring proper design controls, document control, and corrective/preventive actions.
Risk Management: ISO 14971 risk management processes for medical devices, including risk analysis, evaluation, and control measures.
Design Controls: Formal design control processes including design planning, input/output requirements, reviews, verification, and validation.

Documentation and Traceability
Material Certificates: Complete material traceability with certificates of analysis, biocompatibility test results, and regulatory compliance documentation.
Manufacturing Records: Detailed manufacturing records including process parameters, quality control testing, and batch documentation for full traceability.
Change Control: Formal change control procedures for any modifications to materials, processes, or specifications affecting regulatory compliance.
Supplier Qualification: Comprehensive supplier qualification programs ensuring all materials and components meet medical device requirements.
Validation Requirements
Biocompatibility Testing: USP Class VI testing including systemic injection, intracutaneous, and implantation tests to verify biological safety.
Cleaning Validation: Documented cleaning procedures with validation data proving effective removal of contaminants and bioburden reduction.
Sterilization Validation: Validation of sterilization processes including dose mapping, sterility assurance levels, and material compatibility studies.
Performance Testing: Electrical, mechanical, and environmental testing to verify performance meets medical device requirements throughout product lifecycle.
Regulatory Submission Support
510(k) Documentation: Technical documentation supporting FDA 510(k) submissions for medical devices incorporating cable glands.
Technical Files: Comprehensive technical files for CE marking under Medical Device Regulation (MDR) in European markets.
Quality Agreements: Formal quality agreements with suppliers defining responsibilities, specifications, and compliance requirements.
Audit Support: Support for FDA, notified body, and customer audits including documentation review and facility inspections.
What Materials Are Required for Cleanroom Applications?
Cleanroom applications require materials with low outgassing properties, particle generation resistance, chemical compatibility with cleaning agents, smooth non-porous surfaces, and appropriate conductivity levels, with medical-grade stainless steel 316L, PEEK, PTFE, and specialized cleanroom-approved polymers providing optimal performance while meeting ISO 146444 cleanroom standards and maintaining electrical integrity in controlled environments.
Material selection is critical because cleanrooms require strict control of particulate and molecular contamination that could compromise product quality or sterile conditions.
Stainless Steel Requirements
316L Medical Grade: Austenitic stainless steel with low carbon content providing superior corrosion resistance and biocompatibility for medical applications.
Electropolished Finish: Electropolishing removes surface imperfections, reduces particle generation, and creates smooth surfaces for effective cleaning.
Passivation Treatment: Chemical passivation enhances corrosion resistance and removes iron contamination that could cause discoloration or particle generation.
Surface Roughness: Ra values below 0.8 micrometers (32 microinches) minimize bacterial adhesion and facilitate cleaning and sterilization processes.
High-Performance Polymers
PEEK (Polyetheretherketone): Excellent chemical resistance, low outgassing, and biocompatibility make PEEK ideal for pharmaceutical and medical device applications.
PTFE (Polytetrafluoroethylene): Superior chemical inertness and non-stick properties provide excellent resistance to cleaning chemicals and biological materials.
Medical-Grade Nylons: Specially formulated nylons with USP Class VI approval offer good mechanical properties with biocompatibility for medical applications.
Cleanroom-Approved POM: Polyoxymethylene with low particle generation and good dimensional stability for precision cleanroom applications.
Cleanroom Classification Compatibility
ISO Class 5 (Class 100): Ultra-smooth surfaces with minimal particle generation for semiconductor and pharmaceutical manufacturing applications.
ISO Class 6 (Class 1000): Moderate particle control for medical device manufacturing and some pharmaceutical processes.
ISO Class 7 (Class 10000): Standard cleanroom requirements for general pharmaceutical and medical device assembly operations.
ISO Class 8 (Class 100000): Basic cleanroom requirements for packaging and some medical device manufacturing processes.
Outgassing and Contamination Control
Low Outgassing Materials: Materials with minimal volatile organic compound (VOC) emissions that could contaminate sensitive processes or products.
Molecular Contamination: Control of molecular contaminants including silicones, plasticizers, and other organic compounds that could affect product quality.
Ionic Contamination: Materials with low ionic content to prevent contamination of electronic components and pharmaceutical products.
Extractable Substances: Minimal extractable substances that could leach into pharmaceutical products or cleaning solutions during use.
Chemical Compatibility Matrix
| Cleaning Agent | 316L SS | PEEK | PTFE | Medical Nylon |
|---|---|---|---|---|
| Isopropyl Alcohol | Excellent | Excellent | Excellent | Good |
| Hydrogen Peroxide | Excellent | Excellent | Excellent | Fair |
| Quaternary Ammonium | Excellent | Excellent | Excellent | Good |
| Sodium Hypochlorite | Good | Excellent | Excellent | Poor |
| Peracetic Acid | Good | Excellent | Excellent | Poor |
Hassan, who manages operations at a state-of-the-art pharmaceutical facility in Switzerland, needed to upgrade cable glands in their ISO Class 5 cleanroom for sterile injectable manufacturing. The existing glands were generating particles during cleaning cycles and couldn’t withstand the aggressive sterilization protocols required for their new biologic products. We provided electropolished 316L stainless steel cable glands with validated low particle generation and complete chemical compatibility with their cleaning and sterilization processes. The upgrade achieved particle counts 90% below ISO Class 5 limits, eliminated cleaning-related contamination events, and supported successful validation of their new sterile manufacturing line.
How Do You Ensure Proper Cleaning and Sterilization Compatibility?
Ensuring proper cleaning and sterilization compatibility requires selecting materials that withstand repeated exposure to cleaning chemicals and sterilization methods, designing surfaces for complete cleanability, validating cleaning procedures, and maintaining detailed documentation, with medical cable glands requiring smooth crevice-free surfaces, chemical-resistant materials, and validated cleaning protocols that achieve required sterility assurance levels.
Cleaning and sterilization compatibility is essential because inadequate decontamination can lead to product contamination, patient safety risks, and regulatory violations.
Sterilization Method Compatibility
Steam Autoclaving: Materials must withstand 121°C-134°C steam autoclaving5 cycles without degradation, dimensional changes, or seal failure.
Gamma Radiation: Resistance to gamma radiation doses of 25-50 kGy commonly used for medical device sterilization without material degradation.
Ethylene Oxide (EtO): Chemical compatibility with EtO sterilization including resistance to the sterilant and proper degassing characteristics.
Hydrogen Peroxide Plasma: Compatibility with low-temperature plasma sterilization systems including material stability and complete sterilant penetration.
Cleaning Validation Requirements
Cleaning Procedures: Documented cleaning procedures specifying chemicals, concentrations, contact times, and mechanical action required for effective decontamination.
Validation Protocols: Formal validation studies demonstrating cleaning effectiveness using worst-case contamination scenarios and analytical testing.
Acceptance Criteria: Defined acceptance criteria for cleanliness including visual inspection, particle counts, bioburden levels, and chemical residue limits.
Routine Monitoring: Ongoing monitoring programs to verify continued cleaning effectiveness and identify any degradation in performance.
Surface Design Considerations
Drainage Features: Design features that promote complete drainage of cleaning solutions and prevent pooling that could harbor contaminants.
Accessibility: All surfaces must be accessible for cleaning with standard cleaning tools and procedures used in medical facilities.
Disassembly Requirements: Consider whether glands need to be disassembled for cleaning or if intact cleaning is sufficient for the application.
Gasket Replacement: Procedures for gasket replacement and validation when sealing components require periodic replacement due to cleaning chemical exposure.
Documentation and Compliance
Cleaning Instructions: Detailed cleaning instructions including step-by-step procedures, chemical specifications, and safety precautions.
Material Safety Data: Complete material safety information including chemical compatibility, temperature limits, and sterilization compatibility.
Validation Reports: Cleaning validation reports demonstrating effectiveness and establishing routine monitoring requirements.
Training Materials: Training materials for facility personnel on proper cleaning, handling, and maintenance procedures for medical cable glands.
Quality Control Testing
Bioburden Testing: Regular testing for microbial contamination to verify cleaning effectiveness and identify potential problem areas.
Endotoxin Testing: Testing for bacterial endotoxins that could cause pyrogenic reactions in pharmaceutical and medical device applications.
Particle Counting: Particle count testing to verify cleanroom requirements are maintained after cleaning and maintenance activities.
Chemical Residue Analysis: Testing for cleaning chemical residues that could contaminate products or affect biocompatibility.
What Are the Key Selection Criteria for Different Medical Applications?
Key selection criteria vary by application but include regulatory compliance requirements, biocompatibility levels, cleanroom classifications, sterilization methods, chemical exposure, and electrical performance needs, with surgical equipment requiring highest biocompatibility, pharmaceutical manufacturing needing chemical resistance, and diagnostic equipment focusing on electrical integrity while all applications requiring appropriate regulatory certifications and cleaning compatibility.
Different medical applications have unique requirements that must be carefully considered during cable gland selection to ensure optimal performance and regulatory compliance.
Surgical and Patient Contact Equipment
Direct Contact Requirements: USP Class VI biocompatibility with additional cytotoxicity testing for materials in direct patient contact during surgical procedures.
Sterilization Frequency: Ability to withstand frequent sterilization cycles without degradation, typically requiring materials stable through hundreds of sterilization cycles.
Electrical Safety: Enhanced electrical safety requirements including low leakage current and reliable insulation for patient-connected equipment.
Emergency Reliability: Critical reliability requirements for life-support and emergency medical equipment where failure could directly impact patient safety.
Pharmaceutical Manufacturing
GMP Compliance: Good Manufacturing Practice requirements including material traceability, change control, and validation documentation.
Product Contact Safety: Materials that won’t contaminate pharmaceutical products through leaching, particle generation, or chemical interaction.
Cleaning Validation: Extensive cleaning validation requirements with documented procedures and acceptance criteria for pharmaceutical manufacturing.
Batch Documentation: Complete batch documentation and traceability for materials used in pharmaceutical manufacturing equipment.
Diagnostic and Laboratory Equipment
Precision Requirements: Dimensional stability and precision for analytical instruments requiring consistent mechanical and electrical performance.
Chemical Resistance: Resistance to laboratory chemicals, reagents, and cleaning solvents commonly used in diagnostic applications.
EMC Performance: Electromagnetic compatibility requirements for sensitive analytical instruments and diagnostic equipment.
Calibration Stability: Mechanical stability that won’t affect instrument calibration or measurement accuracy over time.
Medical Device Manufacturing
Process Validation: Materials and components that support process validation requirements for medical device manufacturing.
Quality Systems: Integration with ISO 13485 quality systems including supplier qualification and incoming inspection procedures.
Risk Management: Materials that support medical device risk management requirements including failure mode analysis and mitigation strategies.
Regulatory Support: Supplier support for regulatory submissions including technical documentation and regulatory expertise.
Selection Decision Matrix
| Application Type | Primary Criteria | Secondary Criteria | Critical Standards |
|---|---|---|---|
| Surgical Equipment | Biocompatibility | Sterilization | USP Class VI, ISO 10993 |
| Pharmaceutical | Chemical Resistance | GMP Compliance | FDA 21 CFR, cGMP |
| Diagnostic | Precision | EMC Performance | IEC 61326, ISO 15189 |
| Manufacturing | Process Validation | Quality Systems | ISO 13485, 21 CFR 820 |
Cost-Benefit Analysis
Initial Cost vs. Lifecycle: Consider total cost of ownership including maintenance, replacement, and compliance costs over equipment lifetime.
Regulatory Risk: Balance material costs against regulatory compliance risks and potential costs of non-compliance.
Performance Requirements: Ensure selected materials meet minimum performance requirements without over-specification that increases costs unnecessarily.
Supplier Capabilities: Evaluate supplier capabilities for ongoing support, documentation, and regulatory compliance assistance.
Conclusion
Selecting cable glands for medical equipment and cleanrooms requires understanding unique regulatory, material, and performance requirements that distinguish these applications from standard industrial uses. Success depends on balancing biocompatibility, cleanability, regulatory compliance, and electrical performance.
The key to reliable medical cable gland performance lies in working with suppliers who understand medical device regulations and can provide appropriate materials, documentation, and ongoing support throughout the product lifecycle. At Bepto, we specialize in medical-grade cable glands that meet FDA requirements and international medical device standards, providing the quality assurance and regulatory support needed for critical healthcare applications.
FAQs About Medical Cable Glands
Q: What certifications do I need for medical cable glands?
A: Medical cable glands typically require USP Class VI biocompatibility certification, FDA 21 CFR compliance for materials, and ISO 13485 manufacturing quality systems. Specific requirements depend on your application and regulatory jurisdiction.
Q: Can regular stainless steel cable glands be used in medical applications?
A: Regular stainless steel may not meet medical requirements. Medical applications need 316L medical-grade stainless steel with electropolished finish, biocompatibility testing, and proper documentation for regulatory compliance.
Q: How do I clean and sterilize medical cable glands?
A: Follow manufacturer’s validated cleaning procedures using approved cleaning agents and sterilization methods. Most medical glands support steam autoclaving, gamma radiation, or chemical sterilization with proper material compatibility.
Q: What’s the difference between cleanroom and medical grade cable glands?
A: Medical grade includes biocompatibility requirements and FDA compliance while cleanroom grade focuses on particle control and chemical resistance. Some applications require both medical and cleanroom specifications.
Q: How often should medical cable glands be replaced?
A: Replacement frequency depends on sterilization cycles, chemical exposure, and manufacturer recommendations. Typically inspect after every 100-200 sterilization cycles and replace based on condition assessment and validation requirements.
-
Learn about the specific biological reactivity tests required for USP Class VI certification. ↩
-
Access an overview of the ISO 10993 series of standards for the biological evaluation of medical devices. ↩
-
Review the official FDA guidelines and regulations for Current Good Manufacturing Practices (cGMP). ↩
-
Explore a detailed guide to the ISO 14644 standards for classifying air cleanliness in cleanrooms. ↩
-
Understand the principles of steam sterilization, including the role of time, temperature, and pressure. ↩



